Western Exploration Inc. (TSXV: WEX) (OTCQX: WEXPF) (the "Company" or
"Western Exploration") is pleased to provide an update regarding the
initial assay results following the completion of the Company's Phase 1
drill program at Doby George, one of three main mineral deposits located
at the Company's wholly-owned Aura project, located in Elko County,
Nevada (the "Aura Project").
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20221026005363/en/
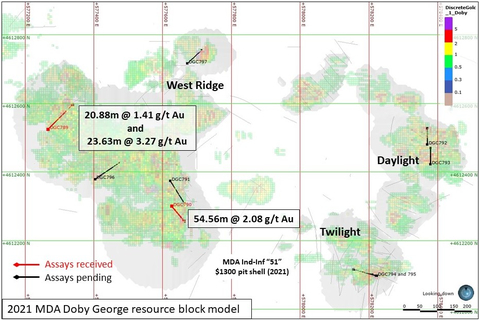
Figure 1. Location of 2022 PQ Metallurgical test holes and resource zones in the Doby George deposit (Graphic: Business Wire)
DRILL HIGHLIGHTS
-
DGC789 intersected 20.88 meters of 1.41 g/t and 23.63 meters of 3.27 g/t Au, including 7.32 meters of 5.27 g/t Au.
-
DGC790 intersected 54.56 meters of 2.08 g/t Au, including 6.4 meters of 4.70 g/t Au.
Darcy Marud, Chief Executive Officer and Director of Western
Exploration, commented: "The first two drill holes completed at Doby
George demonstrate the remarkable continuity and tenor of near surface
oxide gold mineralization and support our assumption that Doby George
can be a high-quality, high-grade oxide gold deposit. Importantly, wide
zones of higher-grade mineralization offer potential to expand the
current resources, not only within the oxide zone but also at depth
within the unoxidized part of Doby George."
The complete results for DGC789 and DGC790 are reported below in Table 1. Assay results for the remaining seven drill holes are pending.
Table 1. 2022 Doby George Drill Hole Results
Hole
|
From
(m)
|
To
(m)
|
Drill Width
(m)
|
Est True Width
(m)
|
AuFA
( g/t)
|
AuCN
(g/t)
|
AuCN/AuFA
Ratio (2)
|
DGC 789
|
63.09
|
83.97
|
20.88
|
19.6
|
1.41
|
1.36
|
97%
|
and
|
89.15
|
112.78
|
23.63
|
22.2
|
3.27
|
2.94
|
90%
|
Including
|
89.15
|
96.47
|
7.32
|
6.9
|
5.27
|
4.67
|
89%
|
and
|
124.05
|
132.44
|
8.38
|
7.9
|
1.09
|
1.02
|
94%
|
DGC790
|
33.83
|
88.39
|
54.56
|
42.0
|
2.08
|
1.88
|
90%
|
Including
|
65.08
|
71.48
|
6.40
|
4.9
|
4.70
|
4.26
|
91%
|
| Note: |
(1)
|
|
Reported at a minimum of 3.0 m @ 0.3 g/t cutoff.
|
(2)
|
|
DGC789 AuCN/AuFA ratios range from 80 to 113%, based on 26 samples;
DGC790 AuCN/AuFA ratios range from 55 to 115%, based on 32 samples, with
only three samples below 80%; all AuCN values were capped at 100% of
the AuFA assays.
|
All sample intervals were assayed by both fire assay (AuFA) and cyanide
soluble shake tests (AuCN) followed up by atomic absorption finish. The
AuCN results are a measure of the solubility of Au in dilute CN
solutions and the state of oxidation of the samples. An AuCN/AuFA ratio
of greater than 80% generally indicates strong oxidation and good
cyanide extraction potential.
DRILL PROGRAM DETAILS
The Phase 1 drill program was comprised of nine large diameter (PQ) core
holes which were completed between July 26, 2022 and September 12,
2022. The drill holes were located at eight different drill sites for a
total of 1,138 metres at the Doby George deposit (see Figure 1 and Table
2 below).
The purpose of the Phase 1 drill program was to evaluate areas within, and adjacent to, the current resource boundary to:
-
validate and delineate current mineral resources;
-
locally test for the expansion potential and structural controls on mineralization;
-
provide samples for metallurgical test work to outline leach kinematics,
as well as mineralized material and waste rock characteristics; and
-
expand upon geotechnical studies through additional test work.
Table 2. Drill Hole Coordinates
Drill Hole
|
Easting
|
Northing
|
Elevation
|
Azimuth
|
Dip
|
Depth (m)
|
DGC789
|
577,267
|
4,612,525
|
2,093
|
45
|
53
|
167.64
|
DGC790
|
577,626
|
4,612,300
|
2,055
|
140
|
55
|
104.24
|
DGC791
|
577,622
|
4,612,374
|
2,058
|
148
|
45
|
109.27
|
DGC792
|
578,370
|
4,612,480
|
2,027
|
0
|
55
|
82.9
|
DGC793
|
578,379
|
4,612,423
|
2,023
|
0
|
55
|
82.29
|
DGC794
|
578,222
|
4,612,097
|
1,966
|
285
|
54
|
118.87
|
DGC795
|
578,221
|
4,612,098
|
1,966
|
285
|
82
|
88.39
|
DGC796
|
577,404
|
4,612,379
|
2,103
|
55
|
69
|
283.47
|
DGC797
|
577,672
|
4,612,716
|
2,039
|
47
|
53
|
100.6
|
Holes DGC789 and DGC790 were completed in the West Ridge resource area
of Doby George, which accounts for approximately 82% of the current
mineral resource estimate at Doby George. The drill holes are
approximately 440 meters apart and showed very positive grade
distribution when compared to the resource block model prepared for the
Company by Mine Development Associates and contained in the technical
report titled "2021 Updated Resource Estimates and Technical Report For the Aura Gold-Silver Project, Elko County, Nevada" dated October 20, 2021 (with an effective date of October 14, 2021) (see Figure 2 and Figure 3 below) (the "Technical Report"). A copy of the Technical Report is available on SEDAR (www.sedar.com) under Western Exploration's issuer profile.
Once assays results have been received for all nine drill holes
completed in connection with the Phase 1 drill program, McClelland labs
will commence certain heap leach and column leach test work with the
previously crushed and assayed samples. The Company expects to receive
the results of all metallurgical test work at the start of Q2 2023.
About Doby George
Doby George is one of the highest-grade, undeveloped open pit heap leach
resources in Nevada and one of three mineral deposits on Western
Exploration's 100% owned Aura Project. The Aura Project is located 32
kilometers north of the Jerritt Canyon mine, which has been in continual
operation since 1981. The deposit is an outcropping, high-grade
sediment-hosted mineral deposit, with multiple zones of mineralization
identified by drilling in a 1.7 x 0.8 km area (see Figure 4 below). The
zone of oxidation extends approximately 150 to 200 meters below surface,
while limited deep drilling has encountered unoxidized gold
mineralization at depths of over 600 meters below surface.
Homestake Mining Company initially discovered the deposit in the
mid-1980s. Since that time, 827 drill holes have been completed,
totaling 115,197 meters of drilling. This includes 74 drill holes,
totaling 22,622 meters that were drilled by Western Exploration LLC
between 1998 and 2017. Metallurgical test work completed on oxidized
material by Homestake Mining Company, Independence Mining Company, and
Atlas Precious Metals Inc., has indicated favorable metallurgy for
standard cyanide heap leach processing. Results from the previous
metallurgical testing completed on 52 bottle rolls and 23 column leach
samples indicate that heap leach gold recoveries approaching seventy
percent can be expected via a crush of 1 to 1.5 inches.
In addition to an established mineral resource estimate (as determined in accordance with National Instrument 43-101 – Standards of Disclosure for Mineral Projects ("NI 43-101")),
the Aura Project has well established infrastructure including
year-round accessibility by highway and county-maintained road and
nearby access to water and electricity (see Figure 5 below). Western
Exploration has exploration, development, and mining rights on 930
hectares of privately owned fee lands through a mineral lease agreement
with a local landowner and may purchase those fee lands at any time for
the development of Doby George.
The 6,000-hectare Aura Project is unique in the junior mining space
insofar as resources have been identified in three different deposits
(Doby George, Gravel Creek and Wood Gulch), as supported by the
Technical Report (see Table 3 below).
Table 3. Updated resource estimates for the Aura Project as contained in the Technical Report
MINERAL RESOURCE(1)
|
|
Indicated
|
Inferred
|
|
Tonnes
|
Au
(g/t)
|
Au
(ozs)
|
Ag
(g/t)
|
Ag
(ozs)
|
AuEq
(g/t)
|
AuEq
(oz.)
|
Tonnes
|
Au
(g/t)
|
Au
(ozs)
|
Ag
(g/t)
|
Ag
(ozs)
|
AuEq
(g/t)
|
AuEq
(oz.)
|
Doby George(2)
|
12,922,000
|
0.98
|
407,000
|
-
|
-
|
0.98
|
407,000
|
4,999,000
|
0.73
|
118,000
|
-
|
-
|
0.73
|
118,000
|
Wood Gulch(3)
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
4,359,000
|
0.66
|
93,000
|
5.80
|
808,000
|
0.74
|
104,543
|
Gravel Creek(4)
|
1,315,000
|
4.73
|
200,000
|
75.0
|
3,169,000
|
5.81
|
245,271
|
2,744,000
|
4.16
|
367,000
|
60.20
|
5,307,000
|
5.02
|
442,814
|
TOTAL
|
14,237,000
|
1.33
|
607,000
|
75.0
|
3,169,000
|
1.43
|
652,271
|
12,102,000
|
1.48
|
578,000
|
15.74
|
6,115,000
|
1.71
|
665,357
|
Notes:
|
(1)
|
|
Au equivalent oz assumes US$1,800 per oz Au and 70:1 ratio of Ag:Au.
|
(2)
|
|
Pit constrained 0.2 g/t AuEq cutoff for oxide, 0.4 g/t Au cutoff for mixed and 1.4 g/t Au cutoff for unoxidized.
|
(3)
|
|
Pit constrained 0.2 g/t AuEq cutoff.
|
(4)
|
|
3.0 g/t Au cutoff.
|
In 2017, Agnico Eagle Mines Limited became a partner of the Company and
has invested over US$14 million in support of the Company's exploration
initiatives. In addition, Agnico Eagle Mines Limited provides technical
expertise to the Company through a joint technical committee with
Western Exploration's management team.
About Western Exploration
Born from a 25-year history of advancing exploration projects in Nevada
as a private company, today Western Exploration is composed of an
experienced team of precious metals experts that aims to lead Western
Exploration to becoming a premiere gold and silver development company
in North America. The Company's principal asset is the 100% owned Aura
gold-silver project, located approximately 120 kilometers/75 miles north
of the city of Elko, Nevada, and includes three unique gold and silver
deposits: Doby George, Gravel Creek, and Wood Gulch. Additional
information regarding Western Exploration and the Aura Project can be
found on the Company's website and on SEDAR (www.sedar.com) under the Company's issuer profile.
Qualified Person Approval
The scientific and technical content in this news release has been
reviewed, prepared and approved by Mark Hawksworth, General Manager of
the Western Exploration's Aura Project, who is a "qualified person"
within the meaning of NI 43-101.
Quality Assurance – Quality Control
Drill program design, Quality Assurance/Quality Control ("QA/QC")
and the interpretation of results is performed by qualified persons
employing a QA/QC program consistent with NI 43-101 and industry best
practices.
Drill core samples were delivered by Major Drilling to Western
Exploration's secure warehouse/logging facility in Mountain City and
washed, photographed and logged by company geologists. Quality control
(QC) samples are inserted at regular intervals in the sample stream,
including blanks and reference materials with all sample shipments to
monitor laboratory performance.
Drill core samples are transported by Neilsen Exploration to McClelland
Laboratory Inc. located in Sparks, Nevada, for sample preparation and
analysis. Each interval is removed from the core box, weighed and
crushed using a jaw-crusher to an approximately 2" top size. The crushed
interval is blended by repeated coning and is quartered to obtain a
one-quarter split for finer crush. Each one-quarter split is crushed to
10 mesh (2 mm) and split to obtain 0.25 – 0.50 kg for pulverization. For
fire assays, the 0.25 kg split is pulverized to >95%-150 mesh
(106µm). Analysis for gold and silver is completed using a 30-gram fire
assay with AA finish. For the CN soluble tests, A 10-gram aliquot of
sample (>95%-106 µm) is leached by shaking in a 50 mL test tube for 1
hour, at ambient temperature and 33.3% solids, using a solution of NaCN
(5.0 gpL) and pH > 11.0 using NaOH. The solution is separated by
centrifuging. Clear solution is analyzed by AA for gold.
Please see below under the heading "Cautionary Note Regarding Forward-Looking Information" for further details regarding risks facing Western Exploration.
For additional information on the Aura Project, please see the Technical
Report, which has been prepared in accordance with NI 43-101, a copy of
which is available on SEDAR (www.sedar.com) under Western Exploration's issuer profile and on Western Exploration's corporate website (www.westernexploration.com).
Cautionary Note Regarding Forward-Looking Information
This news release may contain "forward-looking information" and
"forward-looking statements" within the meaning of the applicable
Canadian and United States securities legislation (collectively,
"forward-looking statements"). These forward-looking statements, by
their nature, require the Company to make certain assumptions and
involve known and unknown risks and uncertainties that could cause
actual results to differ materially from those expressed or implied in
such forward-looking statements. Any statement that involves
predictions, expectations, interpretations, beliefs, plans, projections,
objectives, assumptions, future events or performance (often, but not
always, using phrases such as "expects", or "does not expect", "is
expected", "interpreted", "management's view", "anticipates" or "does
not anticipate", "plans", "budget", "scheduled", "forecasts",
"estimates", "potential", "feasibility", "believes" or "intends" or
variations of such words and phrases or stating that certain actions,
events or results "may" or "could", "would", "might" or "will" be taken
to occur or be achieved) are not statements of historical fact and may
be forward-looking information and are intended to identify
forward-looking information. This news release contains the
forward-looking information pertaining to, among other things: the focus
and results of the Phase 1 drill program; the Doby George deposit being
located in one of the most favourable mining jurisdictions in the
world; Doby George being one of the highest grade, underdeveloped open
pit heap leach resource in northeast Nevada; the accuracy of results
from prior exploration activities conducted at the Aura Project; the key
assumptions, parameters and methods used to estimate the mineral
resource estimate disclosed in this news release; the prospects, if any,
of the Doby George and Wood Gulch-Gravel Creek mineral deposits; future
drilling at the Aura Project; the significance of historic exploration
activities and results; and the ability to expand the existing mineral
resources at the Aura Project. Such factors include, among others, risks
relating to the ability of exploration activities (including drill
results) to accurately predict mineralization; errors in management's
geological modelling; the ability of Western Exploration to complete
further exploration activities, including drilling; property and royalty
interests in respect of the Aura Project; the ability of the Company to
obtain required approvals; the results of exploration activities; risks
relating to mining activities; the global economic climate; metal
prices; dilution; environmental risks; and community and
non-governmental actions. Although the forward-looking information
contained in this news release is based upon what management believes,
or believed at the time, to be reasonable assumptions, Western
Exploration cannot assure shareholders and prospective purchasers of
securities of the Company that actual results will be consistent with
such forward-looking information, as there may be other factors that
cause results not to be as anticipated, estimated or intended, and
neither Western Exploration nor any other person assumes responsibility
for the accuracy and completeness of any such forward-looking
information. Western Exploration does not undertake, and assumes no
obligation, to update or revise any such forward-looking statements or
forward-looking information contained herein to reflect new events or
circumstances, except as may be required by law.
This release uses the terms "mineral resources", "indicated mineral
resources" and "inferred mineral resources". Mineral resources that are
not mineral reserves do not have demonstrated economic viability. You
should not assume that all or any part of measured or indicated mineral
resources will ever be converted into mineral reserves. Further,
inferred mineral resources have a great amount of uncertainty as to
their existence and as to whether they can be mined legally or
economically. On October 31, 2018, the SEC adopted new mining disclosure
rules ("S-K 1300") that are more closely aligned with current industry
and global regulatory practices and standards, including NI 43-101,
although there are some differences in the two standards. Accordingly,
information concerning mineral deposits contain in this release may not
be comparable with information made public by U.S. companies that report
in accordance with S-K 1300.
For additional information with respect to these and other factors and
assumptions underlying the forward‐looking statements and
forward-looking information made in this news release concerning Western
Exploration, see the management information circular dated November 12,
2021, available electronically under Western Exploration's issuer
profile on SEDAR (www.sedar.com).
The forward-looking statements set forth herein concerning Western
Exploration reflect management's expectations as at the date of this
news release and are subject to change after such date. Western
Exploration disclaims any intention or obligation to update or revise
any forward-looking statements, whether as a result of new information,
future events or otherwise, other than as required by law.
Neither the TSX Venture Exchange nor its Regulation Services Provider
(as that term is defined in the policies of the TSX Venture Exchange)
accepts responsibility for the adequacy or accuracy of this news
release. No stock exchange, securities commission or other regulatory
authority has approved or disapproved the information contained herein.

View source version on businesswire.com: https://www.businesswire.com/news/home/20221026005363/en/




